 
The formation of interstitial impurity defect is shown figure.ġ7 Formation of interstitial impurity defect is shown figure.Įxample: Carbon atoms are interstitial impurity atoms that are added to Iron to make Steel. Interstitial impurities are much smaller than the atoms in the bulk matrix. Interstitial defects Definition: When an impurity atom tries to settle in the interstitial space between the parent atoms of the crystal without displacing, then such a defect is known as interstitial defect. The formation of Frankel defects is described in the following figure.ġ6 1.2. In case of Frankel defect also charge neutrality is maintained.ġ5 AgBr, AgCl, ZnS etc., exhibits Frankel defects This pair of defects is known as Frankel defect. Thus, a vacancy and interstitial defects are created. When a +ve ion leave its site and settles in the interstitial position then it creates a vacancy in its position. Definition: When a pair of vacancies is produced at one positive ion site and one negative ion site by replacing positive and negative ions, then this type of defect is call the Frenkel defect.ġ4 Consider the periodic distribution of +ve and –ve ions in an ionic crystal. 
The formation of vacancies is illustrated in the following figure.ġ3 Frenkel defects It is special case of vacancy and interstitial defects in ionic crystals. To maintain charge neutrality, a –ve ion moves to the crystal surface creating a –ve ion vacancy at its site.ġ2 NaCl, CsCl etc., exhibits Schottky defect The formation of +ve ion vacancy results in excess negative charge inside the crystal. In ionic crystals, there are two types of possible vacancies, namely cation (+ve ion) vacancies and anion (–ve ion) vacancies.ġ1 When a +ve ion from the interior of the lattice moves out of the crystal to its surface, then a +ve vacancy is formed at its site. Definition: When a pair of vacancies is produced at one positive ion site and one negative ion site due to absence of positive and negative ions, then this type of defect is call the Schottky defect. They are: (A) Schottky defects and (B) Frenkel defectsġ0 Schottky defects It is special case of vacancy defects in ionic crystals. 
In case of ionic crystals, there are two types of point defects related to vacancies. In most cases, diffusion (mass transport by atomic motion) can only occur because of vacancies. For most crystals, the thermal energy is of the order of 1 eV per vacancy.ĩ The number of vacancies per unit volume of the crystal depends upon temperature. At high temperature atoms are frequently and regularly change their positions leaving empty lattice sites behind their positions. A crystal lattice with vacancy defect is shown in figure.Ĩ Vacancy defects are mainly due to the imperfect packing during the formation of crystal or due to thermal vibrations of atoms at high temperature. Definition: The defects due to the missing atoms at their lattice sites are called vacancies. Vacancies Vacancies are simplest point defects in a crystal which refers to a missing atom at its site. Point defects are of four types: (1) Vacancies (2) Interstitial defects (3) Substitutional defects (4) Electronic defectsħ 1.1. In general, point defects occur in metallic and ionic crystals. A point defect produces strain in a small volume of the crystal surrounding the crystal, but does not affect the perfections distant parts of the crystal.Ħ Point defects are of four types: (1) Vacancies When an atom is missing or an atom is in an irregular place in the lattice structure, the corresponding defects are known as point defects. Point Defects The point defects are one-dimensional defects Volume defects (Three-dimensional defects)ĥ 1. Surface defects (Two-dimensional defects) 4. Line defects (One-dimensional defects) 3. Point defects (Zero-dimensional defects) 2. The defects in crystals may be confined to a point, line, surface and volume.Ĥ The basic classes of crystal defects are:ġ. 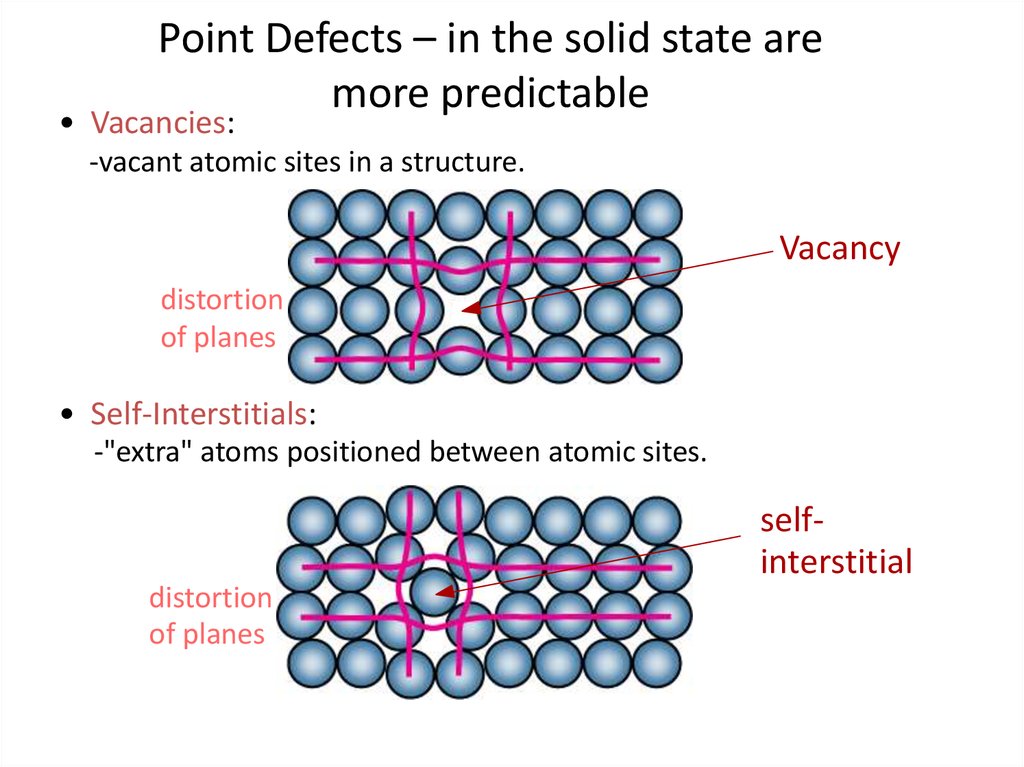
A perfect crystal, with every atom of the same type in correct position, does not exist. Definition: The deviation from the perfect periodicity of atomic arrays in crystals is known as crystal defects.ģ The crystal defects affect their properties such as mechanical strength, ductility, crystal growth, dielectric strength, magnetic hysteresis, conductivity, etc. Any deviation from this periodicity is known as defects or imperfections in crystals. \): Edge Dislocations.Presentation on theme: "Lattice Defects."- Presentation transcript:Ģ INTRODUCTION Crystals will have a regular periodic arrangement of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
